Electropolishing
Upfront assessment of electropolishing process performances
Controlling the quality of metal removal for smoother surfaces
The combination of our in-house CAE solutions and engineering knowledge supports the development of effective electropolishing processes for a variety of components. Whatever the industry sector, from medical to aerospace, or the application, from sterilization to post-production of 3D-printed parts, collaborating with Elsyca offers among other outcomes:
- access insights into process performances including detection of operating window ensuring targeted surface quality (roughness, appearance) objectives;
- validate the performance of the electropolishing solution in combination with the substrate material and define the most optimal process parameters;
- optimize jig/rack design and part layout;
- validate electropolishing cell design, its performance and upscale your production;
- Rely on our expertise to characterize your electropolishing baths.
Predict the microfinish precision of your electropolishing processes by an upfront assessment of the best-operating conditions.
Learn more about our solutions
Did you know?
The process
Electropolishing is a process where a metal substrate (often steel, copper, brass, aluminum or titanium, but also a wide range of other metals and alloys) is immersed in an acid-containing electrolyte solution and an anodic electrical current is imposed onto it. This invokes a controlled corrosion process, effectively removing surface roughness and other surface irregularities, such as sharp edges caused by scratches, or burrs and hair-line surface cracks from preceding manufacturing steps.
Although some electropolishing processes are based on non-aqueous electrolytes or ionic liquids, the vast majority of industrial electropolishing processes rely on an aqueous solution containing phosphoric acid, whether or not in combination with other acids like sulphuric acid or even fluoric acid.
The results
Electropolished surfaces are stress-free and smooth, significantly prolonging the lifetime of the processed device or part. Often electropolished surfaces have a mirrored appearance. the process also improves adhesion through the removal of oils and oxides, yielding a clean surface for any subsequent production step.
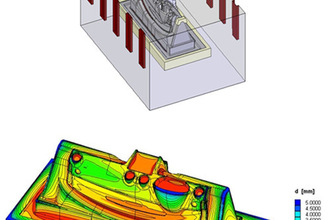
Controlling the current density distribution and electrolyte refreshment (hence the effectiveness of the process) over the substrate is a major challenge for electropolishing processes. Low current density areas might lead to unacceptable roughness values, or even pitting of the substrate. High current density areas might lead to an excessive metal removal rate, thereby compromising the dimensional tolerances of the part, or might even lead to burning in more extreme cases.
The applications
Electropolishing is a surface treatment process that is applied to a huge variety of items, ranging from surgical devices (e.g. stents, needles, tubes) to sensors, valves, storage tanks, fasteners, cutting instruments, gears, shafts to even nuclear accelerators.
Electropolishing provides an effective and efficient method for removing surface roughness and burrs or involuntary sharp edges that remain from preceding manufacturing steps.